Health Highlight: Wed. 26, Apr.
Today’s Highlight(s): A look at some exciting news out of the National Institute of Allergy and Infectious Diseases Vaccine Research Center.
Health Highlights workflow is powered by Glasp.co, Neeva, and Perplexity.ai; produced using iA Writer and Keynote.
Have a question? Want to be a sponsor? Get in touch: carlosfranco.bio
🧠 Learn, ❤️ Like, 🤝 Share, and 📩 Subscribe!

THE BRIEF
In a recent Phase 1 Clinical Trial with 52 healthy volunteers, an experimental flu shot meant to help bring immunity to a range of flu viruses yielded positive results. The vaccine was observed to be safe with minimal side effects and productive in generating antibody responses.

What is new about this vaccine?
What makes this vaccine different is how and to what part the vaccine nanoparticle (H1ssf) binds to the flu virus protein to help the immune system recognize it and begin generating a response.
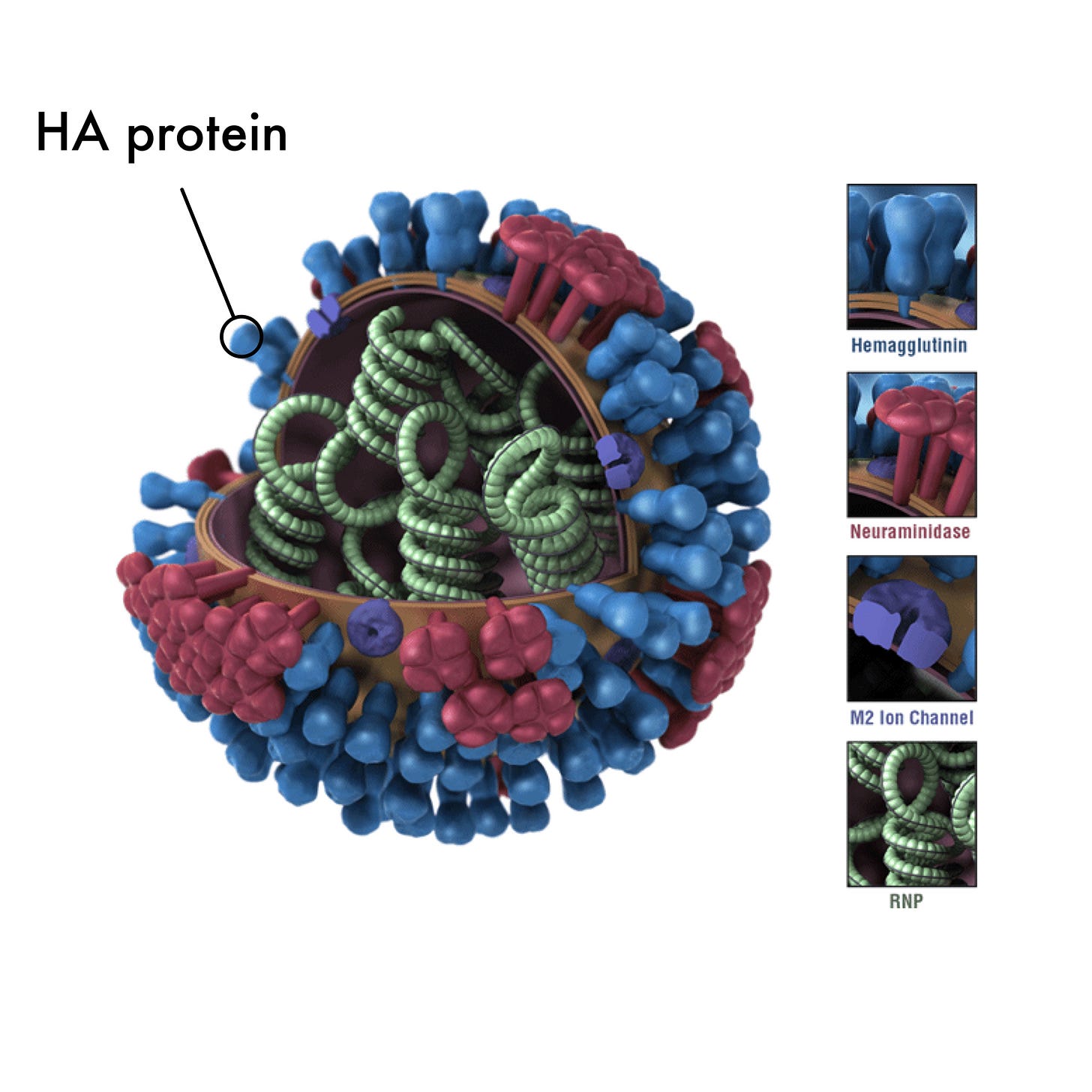
“As anticipated based on preclinical study results, H1ssF generated binding antibodies to the stem of the influenza H1 hemagglutinin (HA) protein.” 1
Context
This type of binding is thought to be advantageous due to the changeability of the HA protein structure at the head. By focusing on less variable and more predictable structures of the stem, researchers hope to generate vaccines that could lead to longer-lasting immunity.
“HA is composed of head and stem domains and enables the influenza virus to attach and enter a human cell. The immune system can mount an immune response to HA, but most of the response is directed toward the head…” 1
Figure 1.
Influenza H1 hemagglutinin (HA) protein2
“The new vaccine candidate consists only of the HA stem. The stem is more conserved than the head between influenza strains and subtypes, and thus is less likely to change every season.” 1
Figure 2.
Model of the H1ssF nanoparticle 1
“The H1ssF vaccine is unique in that it only displays the stem part of the influenza HA protein on the surface of a nanoparticle made of nonhuman ferritin.”
“Ferritin-HA proteins form particles displaying HA spikes on their surface, mimicking the natural organization of HA on the influenza virus. Displaying influenza HA surface proteins on the outside of the nanoparticle makes them easily accessible to immune cells that encounter the nanoparticle.” 1
What’s Next?
Researchers are already working on a second study with the nanoparticle H1ssF, building on what was discovered and used with the SARs-COVID-19 vaccines.
“The H1ssF vaccine using an mRNA delivery system also began testing in a phase 1 clinical trial being overseen by scientists at the Duke Human Vaccine Institute, a part of [National Institute of Allergy and Infectious Diseases] NIAID’s [Collaborative Influenza Vaccine Innovation Centers] CIVICs network.” 1
More
Types of Influenza Viruses [www.cdc.gov]
There are four types of influenza viruses: A, B, C, and D. Influenza A and B viruses cause seasonal epidemics of disease in people (known as flu season) almost every winter in the United States.
What are mRNA vaccines and how do they work? [www.medlineplus.gov]
mRNA vaccines work by introducing a piece of mRNA that corresponds to a viral protein, usually a small piece of a protein found on the virus’s outer membrane.
mRNA vaccines — a new era in vaccinology [www.ncbi.nlm.nih.gov]
This review provides a detailed overview of mRNA vaccines and considers future directions and challenges in advancing this promising vaccine platform to widespread therapeutic use.
Keywords 🏷️: Influenza VaccineResearch ClinicalTrial NIAID-NIHNOTES
“Universal Influenza Candidate Vaccine Performs Well in Phase 1 Trial” https://www.niaid.nih.gov/news-events/vrc-uni-flu-vax
“Types of Influenza Viruses [with HA label]” https://www.cdc.gov/flu/about/viruses/types.htm